The shale gas industry has been trying to build demand for fossil fuels from its fracked oil and gas wells by promoting the construction of a new petrochemical corridor in America’s Rust Belt and expanding the corridor on the Gulf Coast. To help demystify terms like “natural gas liquids” and “cracker plants,” DeSmog has begun building a guide to some of the equipment and terms used in the plastics and petrochemical industries.
This guide, which will expand over time, is intended to serve as an informal glossary of sorts and an introduction to what happens to fossil fuels that are transformed into chemicals, plastics, vinyl, Styrofoam and a variety of other materials.
Petrochemical Production and the Climate
This field guide is part of Fracking for Plastics, a DeSmog investigation into the proposed petrochemical build-out in the Rust Belt and the major players involved, along with the environmental, health, and socio-economic implications.
These fossil fuels have a significant global warming impact of their own. The methane leaks associated with the natural gas drilling and distribution industry are so pronounced that many experts say burning natural gas for electricity is worse for the climate than burning coal.
While hydrocarbons that are used as raw materials for petrochemical products aren’t burned (and therefore don’t release carbon dioxide into the atmosphere), that leaky infrastructure still results in methane pollution. Methane itself is a powerful greenhouse gas, capable of warming the climate 86 times as much as an equal amount of carbon dioxide over the first two decades after it’s released to the atmosphere.
Making petrochemicals also requires a huge amount of energy — some of the largest petrochemical plants like crackers may have their own power plants on site — and that energy comes from burning fossil fuels.
Executives from major oil and gas companies, wary of the impacts that carbon dioxide pollution controls might have on their long-term prospects, have told investors that they see petrochemicals as the place where demand for fossil fuels will continue to grow, even if the world takes serious action on climate change.
THE NATURAL GAS LIQUIDS (NGLs)
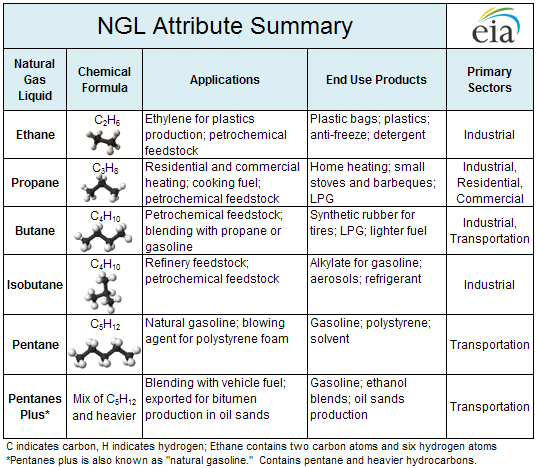
“WET GAS” FROM THE WELL: Oil and gas aren’t the only things that come out of an oil and gas well. There’s also, for example, wastewater and the natural gas liquids, or “wet” fossil fuels, an array of hydrocarbon molecules that flow up to the surface alongside oil, water, and methane, the main ingredient in the natural gas used to heat homes and generate electricity. These “wet” hydrocarbons can represent a surprisingly high proportion of a shale gas well’s production. For every 1,000 cubic feet of methane, some Marcellus Shale wet gas wells can produce up to roughly 9 gallons of natural gas liquids, according to data reported by the American Oil and Gas Reporter in 2011.
Source: The Energy Information Administration
Ethane: Ethane often represents the biggest cut of NGLs from a shale gas well. At room temperature and pressure, ethane is a colorless and odorless gas. Because of its two carbon atoms, ethane is sometimes referred to by the industry as C2.
Ethane “contributes to climate change in three ways,” DeSmog reported in 2016 after researchers found that ethane pollution from the Bakken shale oil field was so severe that it had raised ethane levels in the atmosphere worldwide, “as a greenhouse gas itself (though it’s extremely short-lived in the atmosphere so these effects are relatively fleeting), by extending the lifespan of the powerful greenhouse gas methane (because it consumes compounds that help break methane down), and because it helps to form smog, which the researchers described as ‘the third-largest contributor to human-caused global warming after carbon dioxide and methane.’”
Some drillers leave their ethane mixed in with the methane they sell as natural gas — but natural gas buyers and pipeline companies often set limits on how much ethane can be left in the mix. Once the right infrastructure is built, the petrochemical industry can take ethane and use it to make ethylene (see below).
Propane: Familiar to many from the backyard BBQ grill, propane is the second most-common NGL. It has been marketed as a “green” fuel because it produces less carbon than gasoline or diesel when burned as a fuel for cars and trucks. In 2016, market research firm IHS found that shale drilling was causing U.S. exports of propane to reach record levels. Roughly 6 million homes in the U.S. relied on propane for heating that same year, according to MarketWatch, and the fuel is also widely used in the agricultural industry.
The petrochemical industry can transform propane into propylene (which can then become the plastic polypropylene) as well as ethylene and other petrochemical building blocks. Propane has three carbon atoms per molecule, so it’s sometimes referred to as C3.
Butane: Another colorless, odorless, and flammable gas, butane is the fuel inside cigarette lighters and some camp stoves. Butane burns hot, but it can also be chilled and used as a refrigerant, because it stays in liquid form until temperatures reach 220 degrees below zero (Fahrenheit). It’s also often blended into gasoline for cars and trucks. The petrochemical industry can turn butane into ethylene, as well as acetic acid and the building blocks for resins.
Natural Gasoline/Pentanes Plus: Hydrocarbons with five or more carbon atoms per molecule (C5+) are often referred to in the oil industry as natural gasoline. This mix of NGLs tends to reach its boiling point at or around the same range of temperatures and pressures as gasoline and is often blended into vehicle fuels or used to process tar sands. Pentane, or C5, is also used in the manufacturing of Styrofoam.
The Building Blocks
These materials, sometimes called olefins, are half-way to becoming consumer goods like plastics or chemicals. They’re often produced by “cracking” NGLs (see “crackers” below). While many consumers have never heard their names, they’re the ingredients that form many of the plastic and petrochemical products that wind up on store shelves.
Credit: Plastic Pollution: How Humans are Turning the World into Plastic, a video collaboration with UN Environment and their Clean Seas Campaign, by Kerzegesagt, via YouTube
Ethylene: Ethylene is prized by the petrochemical industry, because it can be turned into polyethylene, Styrofoam, PVC, solvents, ingredients used in shampoos and beauty supplies, and a huge array of other products. Over 60 percent of the ethylene made worldwide ends up as polyethylene — and most polyethylene, in turn, is used to make packaging, according to the American Chemistry Council. Plastic packaging has come under fire from environmentalists, who argue that it makes little sense to use something that is so long-lived for a product that’s designed to be almost immediately discarded. Scientific American reports that single-use plastics and plastic packaging can remain in the environment for over 500 years, causing harm to wildlife, getting into the seafood that people eat, and polluting the world’s oceans.
Propylene: In the petrochemical industry, propylene can be used as a fuel — attractive because it burns hotter than propane — or it can be used as a raw material. Nearly two-thirds of the world’s propylene is turned into the plastic polypropylene, 2007 figures show. The remaining third might wind up in acrylic fibers (sometimes called “artificial wool” and used to make socks, carpets, and more), artificial rubber, ABS plastics (the plastics Lego blocks are made from), or flexible foam found in furniture and automobiles.
Methanol: Although methanol is so often made from wood that it’s also known as “wood alcohol” or “wood spirit,” it can also be made from shale gas. One company, U.S. Methanol, is currently working on dismantling methanol plants in Brazil and Slovenia and moving them to West Virginia, where they will be used to make methanol from shale gas. Methanol is used to make formaldehyde and acetic acid, which in turn can become paints, adhesives, resin, and insulation.
Separating NGLs
Separators and Condensate Tanks: A blend of crude oil, natural gas, NGLs, and wastewater flows up to the surface when an oil and gas well comes online. On the well pad, or at processing stations connected to a group of wellheads, gases and liquids can be separated. Inside separators, gases like methane rise to the top as fluids collect below and are stored in tanks. Tanks near a gas well might hold wastewater (sometimes called brine) or they might hold NGLs in liquid form.
This 2016 FLIR video by environmental group Earthworks shows emissions from condensate tanks at a gas well in Payne County, Oklahoma.
Credit: Earthworks, 2016, via YouTube
Processing Plant: Processing plants can separate out the various hydrocarbons from oil and gas wells more thoroughly than can be done on a well pad. The mix of fluids and gases travels through gathering lines, which are less-regulated, generally smaller-bore pipelines, from the wellhead to a centralized processing plant. At a processing plant, water vapor can be removed through a process called glycol dehydration, which uses a desiccant to absorb water vapor. NGLs can be separated out at processing plants using a similar process or by taking advantage of the fact that each hydrocarbon has different boiling and freezing points, so gas streams can be chilled, heated, or put under differing pressures to isolate NGLs. In the Rust Belt states of Pennsylvania, West Virginia, and Ohio, the U.S. Energy Information Administration (EIA) estimates that gas processing capacity grew nearly 10-fold from 2010 to 2016, when it hit 10 billion cubic feet a day.
Cryogenic Plant: Cryogenic separation is a gas processing method that takes advantage of the fact that methane stays gaseous at very low temperatures, so cryogenic separators chill the combined gases, then send them through a small diameter pipe into an expansion chamber. There, the methane continues flowing as a gas, but most NGLs condense into liquid forms and drop down into the bottom of the expansion chamber.
Fractionation: Fractionators finish the job of separating out NGLs and generally consist of de-ethanizers, depropanizers, debutanizers, and deisobutanizers. As the gas stream passes through towers, where they encounter different pressures and temperatures, a single type of NGL is pulled out of the blend. This Shell Oil video from the 1950s uses a mix of animation and live footage to walk viewers through the science behind the process of separating out different types of hydrocarbon:
Credit: How an Oil Refinery Works Shell Oil Historic Film, by PeriscopeFilm, via YouTube.
In the Rust Belt region, fractional capacity is projected to reach 1.1 million barrels (bbl) a day in 2019, up from 41,000 bbl/day, according to the EIA.
Shipping
NGL Pipeline: Building out an NGL-reliant industry requires the construction of a massive array of pipelines, including the gathering lines that connect wells to processing plants (like the Revolution pipeline that exploded in September 2018, seven days after first going into service) and pipes to carry specific NGLs to buyers, like the Mariner East pipeline project (plagued by construction problems) and Shell’s proposed Falcon pipeline, which will feed its $6 billion ethane cracker plant in Potter County, Pennsylvania. NGL pipelines may also carry ethylene, propylene, and chlorine. The American Chemistry Council estimates that roughly 500 miles of new NGL pipelines passing through Pennsylvania over to Kentucky, will be required to support a new Rust Belt petrochemical corridor.
Compressor Station: At pipeline compressor stations, condensate and wastewater can collect as gas is compressed to propel it across long distances. Those higher pressures can cause liquids to separate out. You may see condensate tanks at these compressor stations.
NGL Export Terminal: When raw ethane and propane are exported for processing overseas, specialized shipping terminals are required to load up liquefied NGLs for transportation by sea. The Marcus Hook terminal in Chester County, Pennsylvania, right outside of Philadelphia, connects to the Mariner East 1 pipeline, and Energy Transfer is currently working on connecting two larger bore pipeline systems, the Mariner East 2 and the Mariner East 2X, to the export terminal as well.
Barge: Cracker plants, which are huge petrochemical complexes, are often built on the banks of rivers, not only because finished products, like tiny plastic beads, can be shipped away by barge, but also because the plants themselves often require pieces of equipment that are too large to transport by road or rail. For example, the de-ethanizer and the C2 splitter at Shell’s Potter Township ethane cracker were both massive, clocking in at a respective 200 feet and 187 feet tall and each weighing hundreds of tons. They arrived at the cracker plant by barge on the Ohio River, as it would have been almost impossible to bring them to the site by road. When companies scout out locations where cracker plants can be built, they must often look for river-side acreage for this reason.
The Gas Station
NGL Storage Site: One of the biggest logistical headaches that the natural gas industry faces is that fact that its product often is, as the name suggests, a gas. Gases are difficult to store and supplies must closely match demand or prices can become fickle. With natural gas liquids, storage can be easier — but to accommodate the volumes of NGLs coming from the Marcellus Shale, the industry still must find huge underground salt formations where companies can store NGLs underground under the conditions to keep them liquid. Once these NGL storage sites are built, they can serve as “gas stations” for ethane crackers — the gigantic plants at the heart of the petrochemicals industry. A single NGL storage site can support multiple crackers. One such site, the Mountaineer NGL storage site, is planned for Monroe County, Ohio. DeSmog has previously reported on the ways that state regulators in Ohio, who have never been faced with permitting a site like Mountaineer, were caught unprepared in what Mountaineer officials called “a relatively undefined setting” for regulation.
Mountaineer, which is expected to cost $500 million, is not the only NGL storage site that experts predict will be hitting drawing boards in the region. In June, an “investment banker/equity capital markets expert who requested anonymity” told Kallanish Energy, a trade publication, that he expected $10 billion in funding for new NGL storage would be available. A $10 billion investment would translate into storage sites with the capacity to store 75 million to 100 million barrels of ethane, ethylene, propane, and propylene, according to the American Chemistry Council.
Transforming Raw Materials
Ethylene Cracker/Ethane Cracker: Cracker plants are huge petrochemical complexes that play a major role in the production of plastics, and they’ve been referred to as the heart of the petrochemical industry. Inside ethylene crackers (which are used to make ethylene), hydrocarbon molecules like ethane or naptha are heated up to temperatures that can reach 1,580 degrees Fahrenheit — as hot as flowing volcanic lava — or higher. At those temperatures, molecular bonds crack apart, and ethylene, propylene, and other byproducts form. These are quickly fed into quenching systems to freeze the cracking process and prevent other unwanted substances from forming. Various contaminants, like tar and coke, must be removed, and propylene and ethylene and other petrochemical building blocks are separated out.
From there, ethylene can be purified, so that sulfur, ammonia, and other impurities are pulled out, and then exposed to a catalyst, causing a chemical reaction that creates polyethylene. That polyethylene can then be shaped into tiny plastic pellets called nurdles (the raw material for producing many plastic products), and hauled away to buyers by truck, rail, or barge.
Often, processing, fractionation, and catalyzation is done on-site at ethane cracker plants. For example, Shell’s ethane cracker under construction in Pennsylvania is designed to accept raw natural gas and transform it into polyethylene all on site.
Historically, many crackers used naphtha, which is derived from crude oil instead of ethane, but with the growth of the shale drilling industry, unwanted ethane supplies have grown, driving prices to historic lows. Crackers that use ethane as a feedstock for ethylene might be referred to as either ethane or ethylene crackers.
“Nine new crackers are expected to come online in the U.S. by 2020 representing 10.7 million tonnes/year of new ethylene capacity,” trade publication Petrochemical Update wrote in October 2018.
Credit: Royal Dutch Shell Ethane Cracker, August 2018, by T. Lynskey, Sr., via YouTube
Propane Dehydrogenation (PDH) Plant: Propane can similarly be turned into propylene, using a process called dehydrogenation. The American Chemistry Council predicts that the Appalachian region can support two new PDH plants, both with the capacity to turn propylene into 490,000 metric tons of polypropylene resin a day.
Downstream Manufacturing Site: Once the building blocks for the petrochemicals industry, like ethylene and propylene, are made, the range of synthetic materials that can be produced from those building blocks becomes enormous. Paints, vinyl, foams, surfactants (or soap-like products), pesticides, and much of the full repertoire of the modern chemicals industry can be formed from the byproducts of the oil and gas industry.
Follow the DeSmog investigative series: Fracking for Plastics
Main image: Petrochemical plant in Saudi Arabia. Credit: Secl, CC BY 3.0
Subscribe to our newsletter
Stay up to date with DeSmog news and alerts